Palladium scavenging: From 1% to within ICH limits
Palladium-catalysed reactions are an increasingly popular method of forming C-C or C-heteroatom bonds. A wide variety of coupling reactions have been developed, which means they can be included in a lot of synthetic pathways. This can allow for complex molecules to be synthesised in fewer stages, which is more efficient and creates less waste. Catalysts can also change the temperature and pressure requirements of a reaction, making a process easier to scale up, an important consideration for the pharmaceutical industry.
The challenge: Meeting ICH guidelines for palladium
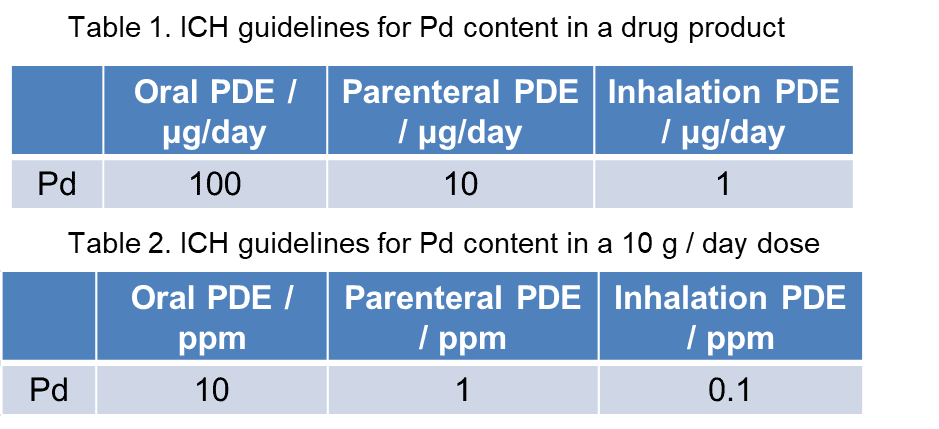
The downfall of palladium catalysts is that drug products must meet the ICH guidelines for palladium content. The permitted daily exposure (PDE) is dependent on the form of administration of the drug and the dosage (Table 1). For an oral medication, the daily dose must have no more than 100 μg of palladium. Table 2 shows how this is applied to a daily dose of 10 g.
To meet these requirements, purification steps such as filtrations, inorganic washes and scavenger treatments are often added to the process. Scavengers can be loaded into a column through which the product can be cycled in a solution, or the free-flowing powder can be added to the solution in a vessel and slurried.
The Onyx approach to palladium-catalysed reactions
Every process responds differently to the various palladium removal techniques, so our development team trials a range of options, such as filtrations, aqueous washes and scavenger treatments, to design a purification method tailored to each project. This method often includes multiple palladium removal steps. Once established, the process is scaled up to 50 L by our non-GMP team to confirm that the large quantities of palladium used in GMP campaigns can still be reduced to within specification.
Case study: Developing an effective palladium removal method
One project involved developing a process to reduce palladium content in material produced using palladium (II) acetate as the catalyst. The aim was to ensure the product met ICH guidelines while maintaining process efficiency during scale-up.
Initial process and early results
The reaction mixture initially contained around 1,000 ppm of palladium. The first method used filtration through a Celite pad to remove the metal. Palladium agglomerates as it moves through processing, and filtration is effective at removing the larger particles. After this and a final purification, which involved a slurry in TBME, the palladium level was reduced to 576 ppm.
Although this showed a reduction in palladium content, further work was needed to reach a more consistent and lower level suitable for GMP manufacture. Considerations for the oxidation state of palladium and further debulking agglomerations that remain ligand bound via tracking ligand removal should be used ahead of resins.
Scaling campaign
For the scale-up campaign, a palladium scavenger treatment was added. The material was slurried with SPM32 overnight before the final purification. Scavengers such as SPM32 have defined pore sizes that allow them to capture the smaller palladium particles that filtration alone cannot remove.
This campaign produced results ranging from 4 to 150 ppm of palladium. Much of the material met the specification depending on dosage and administration route. However, for GMP campaigns, a more reliable and consistent method was required, as lowering palladium content as much as possible gives the client greater flexibility with setting the effective dosage and conforming to regulation standards.
Further process development
Following the scale-up campaign, the process was then developed further to improve reproducibility and achieve lower palladium levels. Several scavengers were tested, including Phos-04 and DMT-functionalised silica, before identifying SEM-26 as the most effective option. Multiple aqueous workups were also assessed, such as sodium diethyldithiocarbamate and N-acetyl cysteine, with N-acetyl cysteine selected for its performance.
The combination of SEM-26 and N-acetyl cysteine formed the basis of the final optimised process, which was tested in a series of stepwise removal experiments.
Stepwise removal and results
Figure 1 shows how the palladium content decreased at each stage of the optimised removal process. The starting reaction contained 151 g of palladium acetate, around 1% of the total mixture.
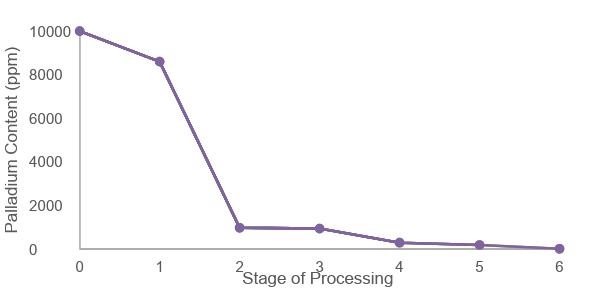
After filtration through a Celite bed (Step 1), the palladium level dropped to 8,600 ppm. This initial step removed a portion of the metal, which had agglomerated into larger particles.
The first N-acetyl cysteine wash (Step 2) had the greatest single impact, reducing palladium to 975 ppm after heating at 30–40°C for 64 hours. A second N-acetyl cysteine wash (Step 3) under the same conditions for 4 hours brought the level down slightly further to 938 ppm. These two steps effectively cleared the larger agglomerates, leaving finer particles in the solution.
Two consecutive SEM-26 treatments followed. The first (Step 4) reduced palladium to 290 ppm after 22 hours at 30–40°C. The second (Step 5), under identical conditions, lowered it again to 185 ppm. These scavenger treatments targeted the smaller particles that filtration and aqueous washes could not remove.
Finally, the TBME slurry purification (Step 6) brought the palladium content to not more than 10 ppm, comfortably meeting the desired specification for this process.
The trend shown in Figure 1 demonstrates a clear and consistent reduction in palladium content through the combination of filtration, aqueous washing and scavenger treatment steps, confirming the effectiveness of the optimised process.
Delivering reliable palladium removal at scale
Onyx Scientific’s development team combines scientific insight with practical problem-solving to overcome complex manufacturing challenges. Through systematic testing, the team refined the palladium removal process to deliver consistent, scalable results that met ICH specifications and supported GMP manufacture.