From lead nomination to commercial supply, our teams will support the development and manufacture of your small molecule drug substance through every stage of its lifecycle.
Our chemist-led, phase appropriate approach ensures the delivery of comprehensive, integrated CMC services, future-proofing against downstream development and production issues.
During 20 years of operation as a small molecule CDMO, a question we’ve often heard from clients is “how do you actually get done as much as you do?”
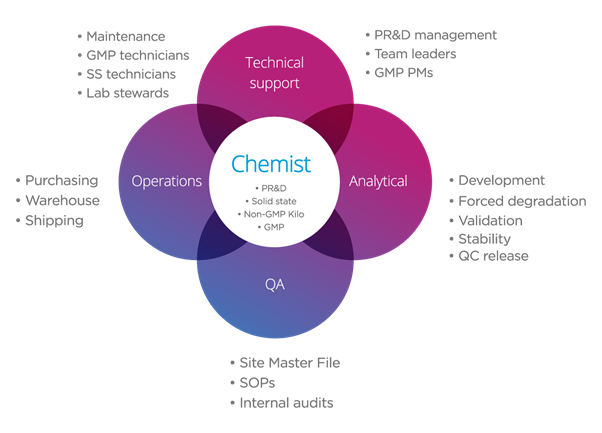
It’s a pertinent question and an observation that reflects what has been the Onyx ethos since day one – to have experts in their fields in the analytical, solid state, operations and quality departments supporting our chemists, who are placed at the centre of everything we do.
This allows our chemists to utilise their skills to the maximum and execute projects without the distraction of those essential supporting activities and results in increased productivity throughout all our projects.
Good chemistry is at the core of everything we do.
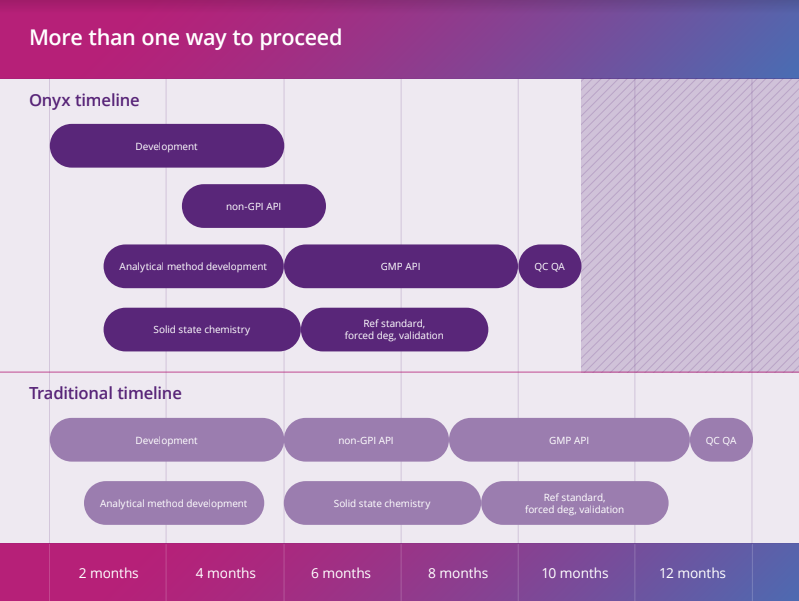
We understand there is no one-size-fits-all approach. At the beginning of a relationship, we take time to understand what is important to you and your project. We construct tailored programmes, outlining possible approaches and striking a balance between risk and reward such that we can deliver a pragmatic, transparent, controlled and suitably risked programme.
We also understand that early-phase work defines the rest of the project, and we deliver fit-for-purpose, phase-appropriate work programs that hit milestone targets, allowing the project to move on to the next stage. We understand what is important now and what can wait until later.
Working to fixed costs and timelines gives a focused approach offering more certainty and better control.
This lean, integrated, chemistry-led approach therefore targets:
● Milestone progression – improving productivity and speed wherever possible
● Risk mitigation – decreasing risk of error and project deviation
● Value – creating lean and efficient processes
● Future-proofing – against downstream development and production issues