Design of Experiments: A systematic approach to process optimisation
Optimising chemical processes is essential to ensuring consistency, efficiency and quality in drug manufacturing.
In this blog, we explore how Design of Experiments provides a more systematic, data-driven way to understand and optimise reactions, define proven acceptable ranges and normal operating ranges and establish a design space that supports robust manufacturing.
The limitations of traditional optimisation approaches
Traditional methods of optimising reactions often involve changing one factor at a time while keeping others constant. Known as one-factor-at-a-time (OFAT), this approach is often used in the early phase of a drug development where producing a sufficient amount of a drug for initial testing is more important than developing an optimal process.
The results of OFAT depend on initial experimental settings and the order of varying the factors. OFAT cannot identify interactions between factors because it only varies one factor at a time. This can lead to incomplete or misleading conclusions, particularly in complex systems where interactions are critical. Nevertheless, data gathered during the OFAT optimisation process creates a knowledge space which is a good starting point for Design of Experiments (DoE) optimisation.
What is Design of Experiments?
DoE is a statistical approach that helps scientists understand the relationships between factors affecting a process and the output of that process. It involves designing a series of experiments in such a way that the effects of multiple variables can be assessed simultaneously.
In organic chemistry, this means identifying the optimal or robust conditions for a reaction, such as temperature, pressure, solvent and catalyst by systematically varying these factors and analysing the results (e.g., yield, purity). A well-designed DoE study can achieve the same level of understanding as OFAT with fewer experiments, saving both time and resources.
The Onyx approach to Design of Experiments
At Onyx, DoE analysis is typically undertaken for Phase 3 or commercial projects. Experimental work is conducted in small, jacketed vessels (Figure 1) designed to replicate our 50 L GMP configuration, ensuring accurate scale-up conditions.

Figure 1. A small, jacketed vessel.
This approach is particularly critical for reactions where vessel geometry plays an important role, such as in hydrogenation. Currently, we maintain a range of vessels with capacities from 0.25 L to 10 L, allowing flexibility in supporting diverse DoE studies. To mitigate variability, all DoE experiments are performed using the same batch of a process-typical material, thereby controlling for any differences in purity.
The DoE process typically follows several key steps:
1. Define the objective
The objective might be to maximize yield or improve the selectivity of a reaction.
2. Identify factors and their ranges
Determine the factors (e.g., temperature, solvent, catalyst charge) that could influence the outcome and choose the ranges at which these factors will be tested.
3. Design the experiment
Common designs include full factorial designs, where all possible combinations of factors and ranges are tested, and fractional factorial designs, which test a subset of possible combinations to reduce the number of experiments.
The relationship between the number of factors (n) and the number of experiments is 2n for a full factorial design. Typically, three repeated experiments are carried out to determine the experimental error.
At Onyx, we prefer to use fractional factorial designs, particularly for experiments with five or more factors, to minimise costs (Table 1).

Table 1. Number of experiments required for full and fractional factorial designs based on the number of investigated factors.
4. Conduct the experiments
The experiments are performed according to the design matrix in an appropriate order. Occasionally, we perform pre-DoE experiments to ensure consistent results with the chosen setup.
5. Data analysis
Statistical software is used to analyse the results, identifying significant factors and their interactions.
6. Optimise
Based on the analysis, the optimal or robust conditions for the reaction, along with the proven acceptable ranges (PAR) and normal operating range (NOR), can be determined. Additional experiments may be required to fine-tune these limits and confirm process consistency. The results are often visualised as a design space, a graphical representation of how process variables interact to affect product quality. Within this space, any combination of variables that meets predefined quality criteria is considered acceptable for routine operation (Figure 2).
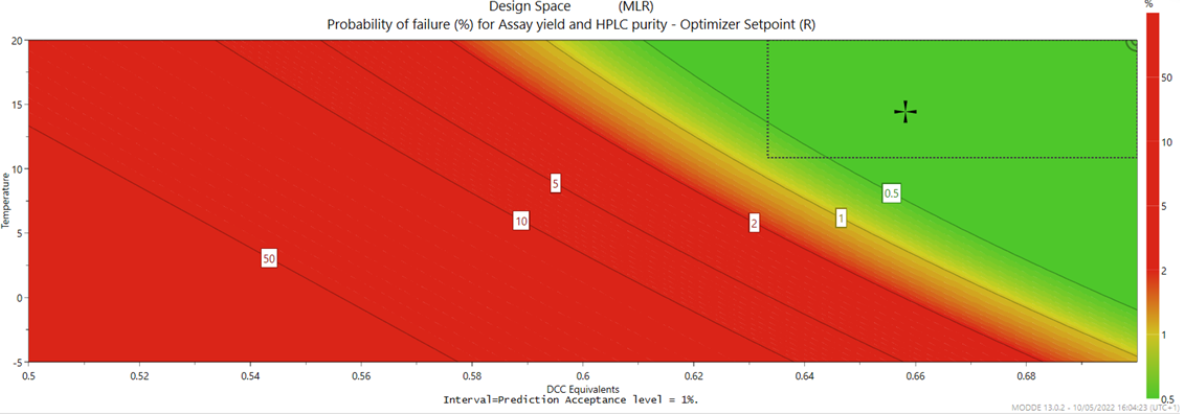
Figure 2. An example of a design space (2D slice).
Defining the proven acceptable range
The PAR defines the boundaries within which the process is known to produce a product that meets all predefined quality attributes (e.g., purity, yield). The PAR can be established by fitting the largest possible regular hypercube within the irregular multidimensional design space volume (illustrated by the dotted frame in Figure 1).
Alternatively, the PAR can be determined for each individual factor while maintaining all other parameters at their designated setpoint values. This latter method is preferred at Onyx, as it is more indicative of real-world scenarios where only one factor is expected to be out of range at any given time.
The PAR ensures that a process can operate safely across a wide range of conditions, providing a buffer for adjustments without compromising quality. Changes within the PAR usually do not require prior approval from regulatory authorities, as these ranges have been validated.
Defining the normal operating range
The NOR maintains day-to-day operational consistency, ensuring that the process stays within a smaller, optimised range to achieve the best possible outcomes. Onyx’s normal operating range in GMP is ±2% around the target value for charges and ±5oC around the target value for temperature.
Establishing and using a design space
Developing a design space helps ensure process robustness by identifying not only the optimal operating conditions but also the boundaries within which the process can run without compromising quality. This leads to enhanced process understanding and control. Design space is not required by authorities to manufacture a product. However, when a design space is approved by regulatory authorities (ICH Q8), operating within the design space is not considered a change and does not require regulatory post-approval changes. This provides flexibility in adjusting process parameters as long as operations remain within the established design space.
7. Process evaluation
At the conclusion of the DoE, the optimised process is evaluated through demonstration batches. This evaluation is typically conducted on a larger scale and performed at least three times to ensure that consistent results are obtained.
Driving smarter process optimisation
While OFAT remains useful for early development, it cannot fully capture the complexity of modern pharmaceutical manufacturing. DoE provides a structured, data-driven approach that delivers deeper process understanding, greater efficiency, and a well-defined design space that supports long-term process control.
At Onyx, our team applies DoE to help clients optimise reactions, establish proven acceptable ranges and design spaces and move confidently toward commercial manufacture.
To learn more about how we can support your process development or validation program, contact our experts today.
